Engineering Software
Brayton Cycle (Propulsion) Analysis
Brayton Cycle -- Propulsion Application
Introduction
This section provides a Brayton Cycle analysis when the working fluid is air.
Analysis
In the presented Brayton Cycle analysis, only air is considered as the working fluid behaving as a perfect gas -- specific heat has a constant value. Ideal gas state equation is valid -- pv = RT.
A gas turbine is a heat engine that uses a high temperature, high pressure gas as the working fluid. Combustion of a fuel in air is usually used to produce the needed temperatures and pressures in the gas turbine, which is why gas turbines are often referred to as combustion turbines. Expansion of the high temperature, high pressure working fluid takes place in the gas turbine. The gas turbine shaft rotation drives an electric generator and a compressor for the working fluid, air, used in the gas turbine combustor. Many gas turbines also use a heat exchanger called a recouperator to impart turbine exhaust heat into the combustor's air/fuel mixture. Gas turbines produce high quality heat that can be used to generate steam for combined heat and power and combined-cycle applications, significantly enhancing efficiency.
The initial state of the working fluid, air, is assumed to be at point 1 at the atmospheric conditions, 298 [K] temperature and 1 [atm] pressure. Air is compressed, isentropically, along line 1-2 by a compressor and enters a combustor. At a constant pressure, combustion takes place (fuel is added to the combustor and the air temperature raises) and/or heat gets added to air. High temperature air exits the combustor at point 3. Then air enters a gas turbine where an isentropic expansion occurs, producing power. Air exits the gas turbine at point 4. It should be mentioned that air at point 1 enters the compressor and the cycle is repeated.
Figure 1 presents a Brayton Cycle schematic layout.
Figure 1 - Brayton Cycle Schematic Layout
Figure 2 presents a Brayton Cycle temperature vs entropy diagram
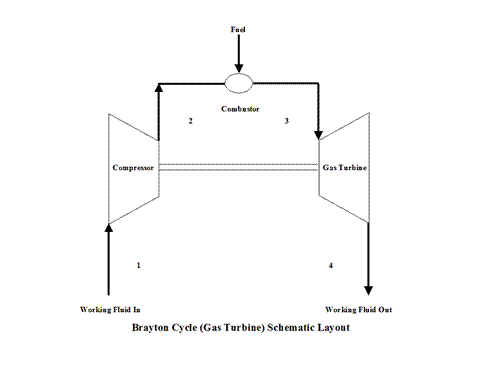
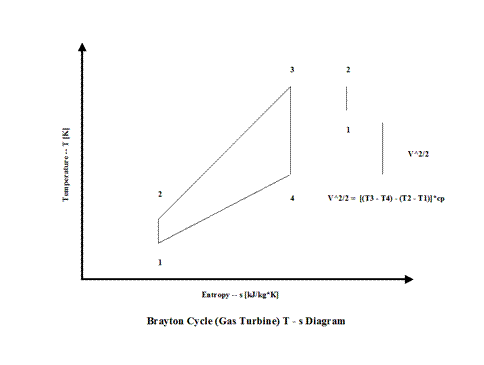
Figure 2 - Brayton Cycle Temperature vs Entropy Diagram
In order to keep the scope of thrust analysis simple, air exiting turbine expands to the atmospheric conditions -- exit pressure is equal to the ambient pressure (p1= p4 ).
It should be pointed out that this material deals with the open Brayton Cycle. The way how the T - s diagram is presented, it describes a closed Brayton Cycle -- this would require a heat exchanger after point 4 where the working fluid would be cooled down to point 1 and the cycle repeats. Therefore, the T - s diagram is presented as a closed Brayton Cycle to allow easier understanding and derivation of the Brayton Cycle thermal efficiency -- heat addition and heat rejection.
The gas turbine and compressor are connected by shaft so the considerable amount of work done on the gas turbine is used to power the compressor.
Propulsion is provided by the difference of gas turbine expansion minus the compressor power requirements.
It can be noticed from the T - s diagram that the work done on the gas turbine is greater than the work necessary to power the compressor -- constant pressure lines in the T - s diagram diverge by going to the right side (entropy wise).
Figure 3 presents the Brayton Cycle efficiency as a function of the compression ratio. It should be noted that the inlet conditions are standard ambient conditions: temperature of 298 [K] and absolute pressure of 1 [atm].
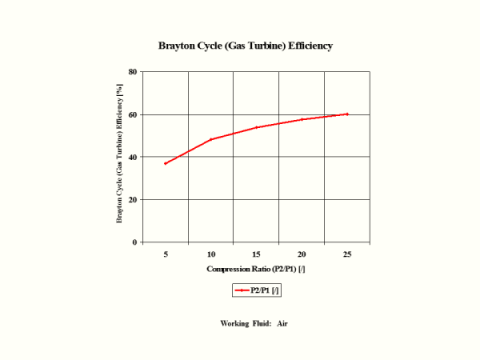
Figure 3 - Brayton Cycle Efficiency
Here, two general performance trends are considered. First, impact of the gas turbine inlet temperature and compression ratio on the Brayton Cycle specific propulsion output and second, impact of the working mass flow rate for a fixed gas turbine inlet temperature on the Brayton Cycle propulsion output.
Figure 4 presents the results of the first performance trend, while Figure 5 presents the results of the second trend.
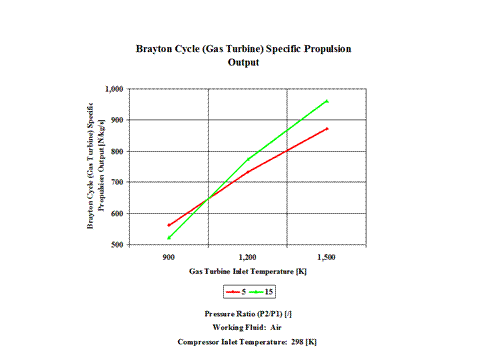
Figure 4 - Brayton Cycle Specific Propulsion Output
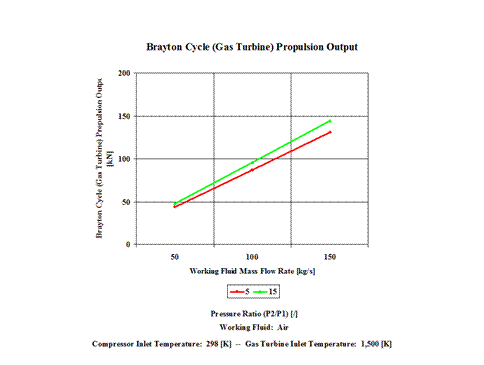
Figure 5 - Brayton Cycle Propulsion Output
One can notice that the Brayton Cycle efficiency increases with an increase in the compression ratio values. One can notice that the Brayton Cycle specific power output increases with an increase in the gas turbine inlet temperature. Furthermore, the increase is greater for the higher compression ratio values.
One can notice that the Brayton Cycle propulsion output increases with an increase in the working fluid mass flow rate for a fixed gas turbine inlet temperature. The increase is greater for the higher compression ratio values.
Assumptions
Working fluid is air. There is no fricion. Compression and expansion processes are reversible and adiabatic -- isentropic. Ideal gas state equation is valid -- pv = RT. Air behaves as a perfect gas --specific heat has a constant value.
Governing Equations
T2/T1 = (p2/p1)^(ϰ-1)/ϰ
p2/p1 = (T2/T1)^ϰ/(ϰ-1)
T3/T4 = (p3/p4)^(ϰ-1)/ϰ
p3/p4 = (T3/T4)^ϰ/(ϰ-1)
ϰ = cp/cv
cp - cv = R
pv = RT
w = qh - ql
qh = cp(T3 - T2)
ql = cp(T4 - T1)
w = cp(T3 - T2) - cp(T4 - T1)
W = (cp(T3 - T2) - cp(T4 - T1))m
efficiency = 1 - 1/rp^(ϰ-1)/ϰ
rp = p2/p1
v^2/2 = cp((T3 - T2) - (T4 - T1))
v = (2cp((T3 - T2) - (T4 - T1)))^1/2
Thrust = vm
Input Data
T1 = 298 [K]
p1 = 1 [atm]
T3 = 900, 1,200 and 1,500 [K]
p3 = 5, 10, 15, 20 and 25 [atm]
R = 0.2867 [kJ/kg*K]
cp = 1.004 [kJ/kg*K]
ϰ = 1.4 [/]
m = 50, 100 and 150 [kg/s]
Results
Brayton Cycle Efficiency vs Compression Ratio
Compression
Ratio
[/]
5
10
15
20
25
Brayton Cycle
Efficiency
[%]
36.92
48.22
53.87
57.53
60.16
Specific Propulsion Output vs Compression Ratio
for a few Gas Turbine Inlet Temperature Values
Specific
Propulsion
[N/kg/s]
Compression Ratio
[/]
5
15
Gas Turbine Inlet
Temperature
[K]
900
563
524
1,200
734
774
1,500
872
961
Propulsion Output vs Compression Ratio
for a few Mass Flow Rates
Gas Turbine Inlet Temperature = 1,500 [K]
Propulsion Output
[kN]
Compression Ratio
[/]
5
15
Mass Flow Rate
[kg/s]
50
43.61
48.06
100
87.23
96.11
150
130.85
144.17
Conclusions
The Brayton Cycle efficiency depends on the compression ratio and working fluid properties. The efficiency increases with an increase in the compression ratio values. Also, the efficiency increases with the higher value for ϰ, which is a ratio of gas specific heat values (cp/cv).
The Brayton Cycle specific propulsion increases with an increase in the gas turbine inlet temperature. Furthermore, the increase is greater for the higher compression ratio values. The Brayton Cycle propulsion increases with an increase in the working fluid mass flow rate for a fixed gas turbine inlet temperature. The increase is greater for the higher compression ratio values.
References
JANAF Thermochemical Data - Tables, 1970